Bomb Calorimetry Testing

Bomb Calorimeter
Parr Bomb calorimetry is used to measure heats of combustion. This includes flammable aerosol cans, aerosol sprays, foodstuffs, aviation fuels, renewable energy sources, explosives, organics, pharmaceuticals, pyrotechnics, and building or construction materials. Fuel and aerosol measurements are based on ASTM D 240-19, Standard Test Method for Heat of Combustion of Liquid Hydrocarbon Fuels by Bomb Calorimeter, or the precision method, ASTM D 4809-18. Building and construction material evaluations are based on the International Standard, ISO 1716: 2018(E), Reaction to fire tests for products- Determination of the gross heat of combustion (calorific value).

Bomb Accessories
The Parr bomb is usually calibrated by combustion of a high purity sample of benzoic acid (NIST SRM 39j or equivalent). For high-precision calorimetry, the mass of the benzoic acid used for calibration will be corrected for buoyancy. After a valid calibration, standard enthalpies of formation for pure compounds may be derived from the standard heats of combustion. For high-precision evaluations, combustion data requires correction for a series of factors collectively known as the “Washburn reduction.” Details of these corrections are described in “EXPERIMENTAL THERMOCHEMISTRY Measurement of Heats of Reaction” edited by Frederick D. Rossini, INTERSCIENCE PUBLISHERS INC. NEW YORK, 1956.

Parr Bomb in the Combustion Bucket
The bomb is fired in a bucket containing 2 kg distilled water. Heat of combustion is calculated from the change in the water temperature.
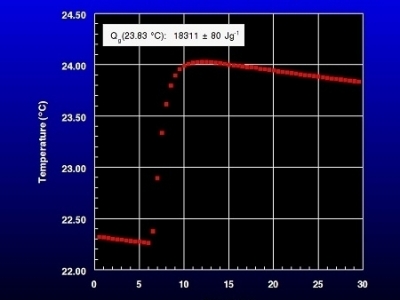
Biomass Combustion
Native Anmerican grasses are a source of renewable energy. Bomb calorimetry is used to evaluate the energy content. The graphic shows the typical combustion curve obtained for an American prairie grass.
